Research Area
Exotic Heterocycles
We design heterocycles that look unfamiliar at first glance, then ask whether that unfamiliarity can be converted into useful reactivity.
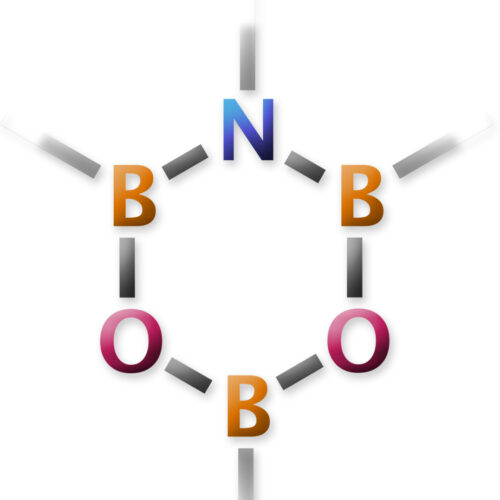
Overview
One central molecule in this program is DATB, 1,3-dioxa-5-aza-2,4,6-triborinane. It is a six-membered heterocycle with no carbon atoms in the ring, yet it behaves as a remarkably practical catalyst for direct amidation from carboxylic acids and amines.
The point is not simply to make an unusual ring. The ring framework positions boron, nitrogen, and oxygen atoms so that carboxylic acids can be activated catalytically, allowing amide bonds to form with water as the main byproduct. That is the sort of chemistry we like: structurally odd, mechanistically rich, and useful enough to matter.
DATB is also thermally robust and has moved beyond a curiosity; a Pym-DATB derivative is commercially available. We are now extending this idea toward broader amidation chemistry and toward new boron-containing ring systems with functions that are still hiding in the periodic table.
What we are asking
Clean amidation
Can a designed heterocycle replace stoichiometric coupling reagents in amide synthesis?
Lewis acidity by design
How does local protonation or ring electronics reveal reactivity that is not obvious from the static structure?
New heteroatom frameworks
What happens when carbon is no longer the default element of an organic ring?
Visual Notes


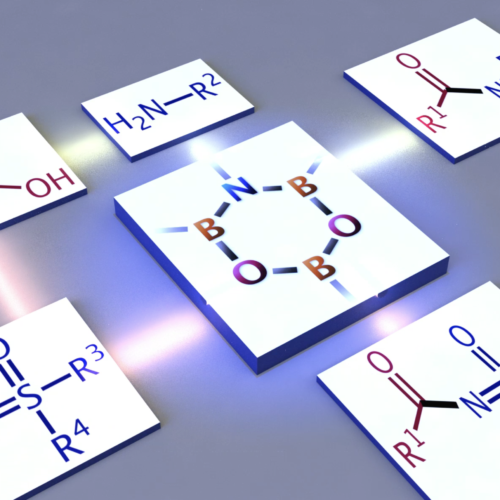
Representative Publications
- Unique Physicochemical and Catalytic Properties Dictated by the B3NO2 Ring System
Hidetoshi Noda, Makoto Furutachi, Yasuko Asada, Masakatsu Shibasaki,* and Naoya Kumagai*
Nature Chem. 2017, 9, 571-577. - Direct N-Acylation of Sulfoximines with Carboxylic Acids Catalyzed by the B3NO2 Heterocycle
Hidetoshi Noda, Yasuko Asada, Masakatsu Shibasaki,* and Naoya Kumagai*
Chem. Commun. 2017, 53, 7447-7450. - All Non-Carbon B3NO2 Exotic Heterocycle: Synthesis, Dynamics, and Catalysis
Christopher R. Opie, Hidetoshi Noda, Masakatsu Shibasaki,* and Naoya Kumagai*
Chem. Eur. J. 2019, 25, 4648-4653. - Expeditious Access to the B3NO2 Heterocycle Enabling Modular Derivatization
Ryosuke Tsutsumi, Nobuaki Kashiwagi, and Naoya Kumagai*
J. Org. Chem. 2023, 88, 6247-6251.